Medannex is an award-winning clinical-stage Scottish biopharmaceutical company committed to unlocking the therapeutic potential of targeting annexin-A1.
“Annexin-A1 may prove a very significant clinical target, and blocking its activity with MDX-124 could represent a completely new and exciting way to treat cancer. We are delighted to be leading the first-in-human study.”
Professor Daniel Palmer,
Liverpool Clinical Trial Centre
We are pioneering the targeting of annexin-A1 to provide powerful new treatment options for people impacted by cancers and autoimmune diseases.
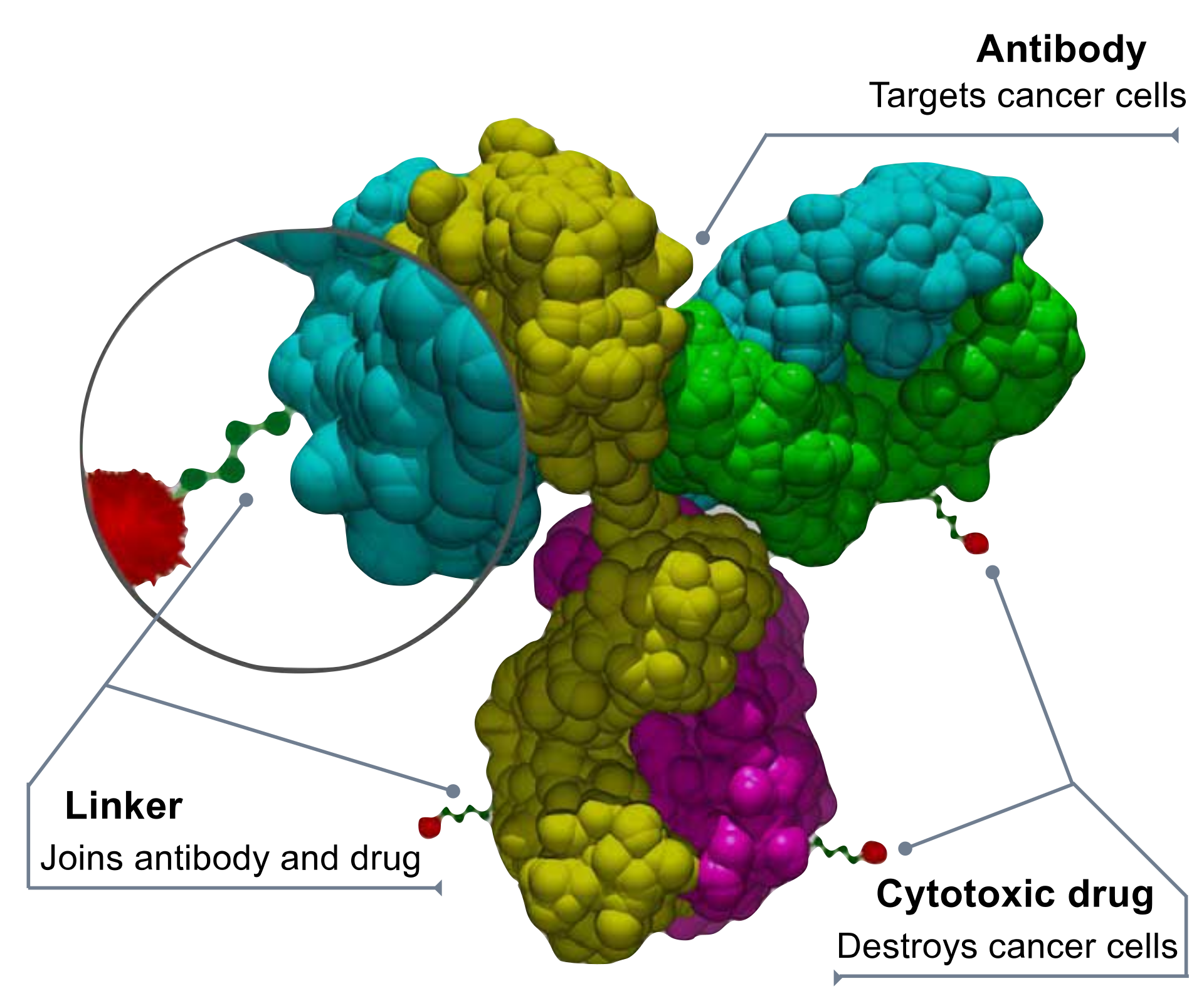
Annexin-A1 drives cancer cell growth and migration and also plays a key role in the development of several autoimmune conditions. Extensive pre-clinical data demonstrate that our first-in-class monoclonal antibody (MDX-124) effectively blocks the action of annexin-A1.
A Phase 1b clinical study is underway, in cancer patients. In parallel, we are currently preparing for further clinical studies in autoimmune diseases.
Medannex is collaborating with world-leading clinical experts to explore the huge therapeutic and commercial potential of our patent-protected technology.